Importance of MDR-compliant Medical Technology
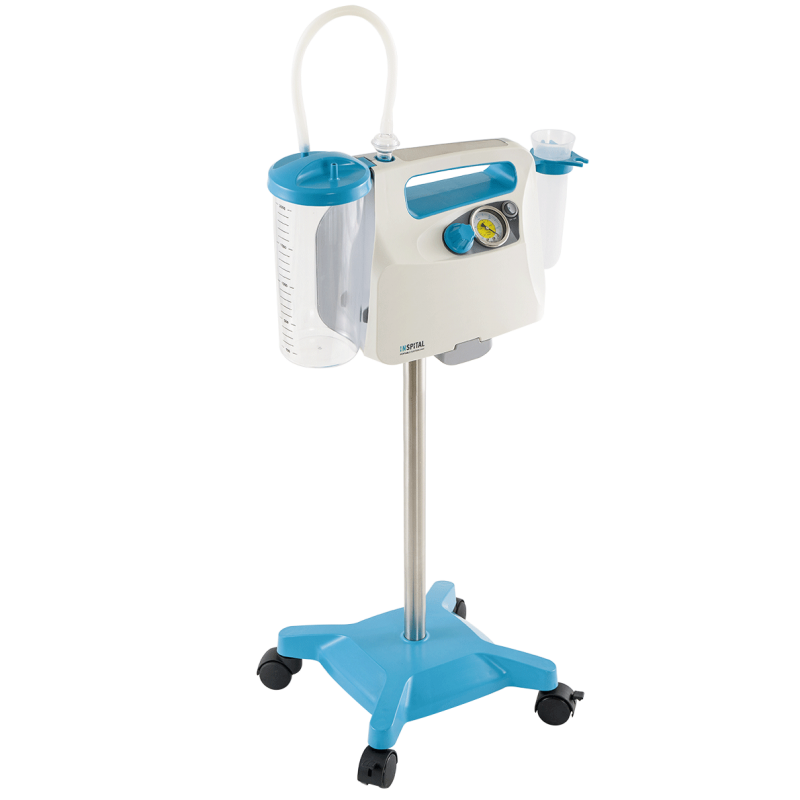
Surgical Suction Machine SU60.05
Prices visible to registered customers onlySurgical Suction Unit SU60.10
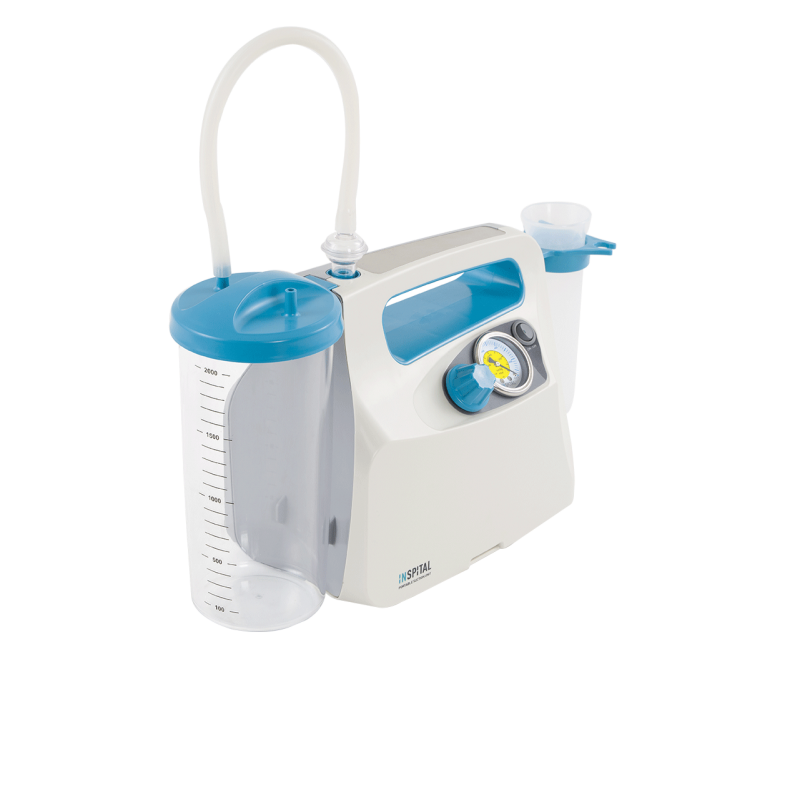
Prices visible to registered customers onlyPortable Suction Unit SU60.06T
Prices visible to registered customers onlyPortable Suction Unit SU60.06
Prices visible to registered customers onlyCeiling supply unit with double joint FX40.20V
Prices visible to registered customers onlyDual-head surgical lights with camera and arm-mounted 27″ medical monitor LD20.53S
Prices visible to registered customers onlyDual-head surgical lights with camera and wall-mounted 27″ medical monitor LD20.24S
Prices visible to registered customers onlyDual-Head Surgical Light LD10.02S
Prices visible to registered customers onlyThree-Headed Surgical Light LD10.03S
Prices visible to registered customers onlySingle-Head Surgical Light LD10.01S
Prices visible to registered customers onlyCeiling service unit with single joint FX40.10V
Prices visible to registered customers onlySingle washbasin SK61.10
Prices visible to registered customers onlyDouble Sink SK61.20
Prices visible to registered customers onlySingle Scrub Sink SK60.10
Prices visible to registered customers onlyDouble Scrub Sink SK60.20
Prices visible to registered customers onlyInstrument Trolley AB00.21
Prices visible to registered customers onlyInstrument Trolley AB00.22
Prices visible to registered customers onlyInstrument Trolley AB00.23
Prices visible to registered customers onlyHydraulic Mayo Table MS10.51
Prices visible to registered customers onlyMechanic Mayo Table MS10.50
Prices visible to registered customers onlyPortable Suction Unit SU60.08
Prices visible to registered customers onlyMobile surgical light with battery LD05.06
Prices visible to registered customers onlyMobile surgical light, satellite head with battery LD05.06S
Prices visible to registered customers onlyDouble Motorized Pendant FX40.25M
Prices visible to registered customers onlyThe MDR (EU) 2017/745: A New Standard for Medical Technology from Neuss
The introduction of the Medical Device Regulation (MDR) marks the most significant turning point in European medical device legislation in decades. For hospitals and medical technology purchasers, the question is no longer whether to transition, but how to ensure compliance. Inspital GmbH, based in Neuss, recognized this transformation process early on as a strategic opportunity. While the old Medical Device Directive (MDD) primarily focused on placing products on the market, the MDR places the entire lifecycle of a product at its core. For our customers in the DACH region, this means a drastic increase in patient safety. In Neuss, we develop and validate our operating tables and surgical lights according to these strict standards to ensure that every clinical application stands on a solid regulatory foundation.
The complexity of the MDR arises from the stricter requirements for technical documentation and clinical evaluation. Every product leaving our facility in Neuss undergoes a meticulous testing process. For clinic operators, this means a significant reduction in liability. When you invest in medical technology that is “Made in Germany” and fully MDR-compliant, you minimize the risk of regulatory complaints from health authorities. Inspital acts not only as a supplier but as a partner, making the regulatory hurdles for Class I and higher-classified products transparent. We understand that clinics in the Rhein-Kreis Neuss and beyond need planning certainty. Therefore, we only offer solutions that are fully marketable beyond 2026. This protects your investments and guarantees continuous patient care at the highest level.
EUDAMED and UDI: Digital Transparency for the Modern OR
A central search term in the context of the MDR is the EUDAMED database. This European database for medical devices serves to enhance transparency and improve market surveillance. Closely linked to this is Unique Device Identification (UDI). Every medical device from Inspital GmbH, from complex ceiling supply units to surgical suction devices, is equipped with a unique UDI code. In Neuss, we have digitized our logistics chains and production processes so that every component is fully traceable. For medical technicians in hospitals, this means enormous efficiency gains in asset management. By scanning the barcode, all relevant safety data sheets, declarations of conformity, and maintenance intervals can be accessed immediately.
This is particularly important for compliance with the Medical Device Operator Ordinance (MPBetreibV). Clinics are obliged to maintain an inventory list, which must be even more detailed under the MDR. Inspital GmbH provides the necessary data packages directly. Our experts in Neuss support you in seamlessly integrating this data into your clinical IT systems. Digital traceability also ensures that in the event of a corrective action (recall), only the affected batches need to be identified, instead of shutting down the entire operation. This saves time, money, and conserves the personnel resources of your technical department. Due to the geographical proximity of our Neuss location to the major metropolitan areas in NRW, we can also offer on-site service that complements digital documentation with physical expertise. Transparency is not an end in itself, but the basis for trust between manufacturer, operator, and patient.
Post-Market Surveillance (PMS): Quality Assurance Far Beyond the Purchase
The MDR requires manufacturers like Inspital to play an active role in Post-Market Surveillance (PMS). This means that we must continuously monitor the performance and safety of our products even after installation in the operating room. At our headquarters in Neuss, we systematically collect and analyze feedback from clinical practice. Whether it’s the ergonomics of our motorized operating tables or the light intensity of our LED systems – every piece of feedback from our users flows directly into our continuous improvement process. This proactive market surveillance is a core component of the MDR and distinguishes reputable manufacturers from cheap importers who are often unreachable after the sale.
For clinic operators, this process represents an enormous gain in safety. They directly participate in technological advancements and can be sure that potential risks are identified before they lead to an incident. Inspital regularly conducts user surveys and post-market clinical follow-ups (PMCF). In Neuss, we evaluate this data to constantly refine the intended purpose of our devices. This is particularly important for products used in sterile environments, such as our stainless steel furniture or suction systems. The MDR requires scientific evidence of clinical utility. By undertaking this effort in Neuss, we provide you with the security you need for your certifications (e.g., according to KTQ or ISO). An Inspital product is therefore never “finished” but continuously evolves with the demands of modern surgery and the regulatory landscape.
Operating Tables and Surgical Lights: High-Performance Technology Under MDR Scrutiny
At the center of every surgical procedure are the operating table and the surgical lights. Under the MDR, these devices are classified as critical components that must withstand the highest stresses. Inspital GmbH manufactures systems in Neuss that are specifically tailored to these requirements. For our operating tables, we focus on radiolucency and mechanical stability for bariatric patients. Every adjustment – whether Trendelenburg or lateral tilt – must be precise and safe under load. The MDR requires extensive load tests and a detailed risk analysis of the control software. In Neuss, we ensure that our EMC tests (electromagnetic compatibility) go far beyond standard norms to exclude interference with other life-sustaining devices in the OR.
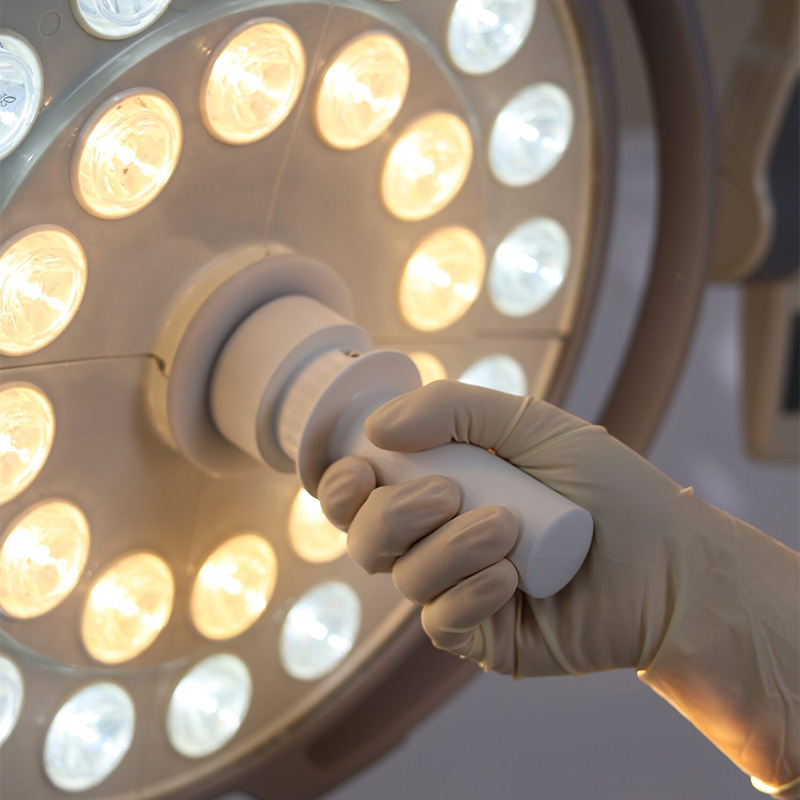
Our LED surgical lights are another example of MDR-compliant precision. Light is a safety factor in the OR. Our systems offer shadow-free illumination and natural color rendering (Ra > 95), which is essential for tissue differentiation. The regulatory requirements for heat development at the patient’s head have been further strengthened under the new regulation. The cooling management systems we develop in Neuss guarantee that the LED arrays remain cool even during hours of surgery and do not disturb the sterile air stratification (TAV ceilings). If you choose Inspital lights, you are investing in technology that is clinically validated and whose performance data has been confirmed by independent testing bodies. This gives surgeons the confidence they need for complex procedures and purchasing departments the assurance of having acquired a future-proof premium product that represents Neuss as a center for medical technology.
Ceiling Supply Units (CSU): Safety and Ergonomics at the Interface
The planning of ceiling supply units is one of the most complex tasks in the construction or renovation of operating rooms. These systems bundle medical gases, electricity, and data lines in a confined space. According to the MDR, these units are often considered a combination of medical devices, which places special demands on conformity assessment. The FX series from Inspital, developed and designed in Neuss, solves this problem with a modular design that is compliant from the outset. We ensure that all interfaces – from the gas outlet to the monitor arm – comply with the strict standards for medical device combinations. This is particularly important for clinics in Germany to avoid unpleasant surprises during acceptance by TÜV or Dekra.
A key factor of the MDR is usability. Our ceiling pendants are designed to be intuitive to operate, virtually eliminating operating errors. In Neuss, we simulate OR workflows to optimize the positioning of the supply arms. This reduces stress for personnel and increases patient safety. The cable-free floor space achieved by our CSUs also indirectly contributes to infection prevention, as cleaning cycles can be carried out more efficiently. Inspital offers a full service from the initial CAD sketch to assembly and maintenance. Due to our location in Neuss, we can react extremely flexibly to construction projects in the Rhineland and nationwide. We coordinate the interfaces with other trades and ensure that your medical technology goes into operation on time and in compliance with MDR.
Stainless Steel Furniture for CSSD: Hygiene as a Regulatory Obligation
The reprocessing of medical devices in the CSSD (Central Sterile Supply Department) is under particular scrutiny under the MDR. Here, stainless steel furniture plays an often underestimated but critical role. Inspital manufactures high-quality stainless steel furniture solutions in Neuss from stainless steel 1.4301 (AISI 304), specifically developed for the aggressive cleaning and disinfection cycles in hospitals. According to the MDR, all materials that come into contact with medical devices must be biocompatible and corrosion-resistant. Our instrument tables, packing tables, and cabinets are “Hygienic by Design.” This means: no open joints, smooth surfaces, and seamless welds to prevent germ colonization (biofilms).
In Neuss, we ensure that our stainless steel products are not only functional but also ergonomic. Height-adjustable work tables in the CSSD protect the health of your employees and increase concentration when checking instrument trays. Under the MDR, the entire process chain of sterile supply is audited more strictly. With Inspital equipment, you meet the highest requirements for hygiene and documentation capability. We provide detailed cleaning validations and material certificates for our furniture. This is a decisive advantage during inspections by the health authorities or the district government of Düsseldorf. Our customers appreciate the longevity of Inspital products. In an age of throwaway mentality, we in Neuss focus on sustainability through quality. Stainless steel is 100% recyclable and remains a safe component of your clinical routine for decades – a direct contribution to economic efficiency and the fulfillment of MDR sustainability aspects.
Strategic Procurement in the MDR Era: Why Inspital Neuss is Your Partner
The procurement of medical technology has fundamentally changed due to the MDR. It is no longer just about the lowest price, but about long-term availability and legal certainty. Many small manufacturers had to cease production due to the high costs of MDR certification. Inspital GmbH, however, has invested massively in the Neuss location and in regulatory compliance. If you buy an operating table or a suction unit from us today, you are choosing investment security. We guarantee the supply of spare parts and software updates over the entire lifecycle of the devices. This is a decisive factor for the risk management of any hospital management.
Our geographical location in Neuss, in the heart of the Rhine-Ruhr metropolitan region, enables us to maintain unique customer proximity. We understand the specific requirements of the German healthcare market and regional legislation. Our consultants come directly to your clinic to develop tailor-made solutions that fit precisely into your budget and schedule. The MDR is a challenge, but with the right partner by your side, it becomes an opportunity for a wave of modernization that will bring your clinic to the technological forefront. Trust in German engineering and the reliability of an owner-managed company from Neuss. Together, we will design an operating room that not only complies with current laws but also sets standards in surgery for tomorrow. Contact us for a non-binding consultation and learn more about our MDR roadmap and our innovative product portfolio.