MDR 2026: A Guide to Modern Hospital Equipment from Inspital
- Current News, Hospital Management, Suction Units

Contents
For Inspital GmbH as a partner for high-quality operating room solutions, one thing takes priority: the symbiosis of the highest technological innovation and uncompromising regulatory conformity. In this article, we examine why the MDR is not merely a bureaucratic obstacle, but a quality seal that places patient safety at the center—and how Inspital supports you in this transition. In addition, it should be noted that the MDR not only re-evaluates existing products, but also influences innovation cycles through stricter documentation requirements. Hospitals must therefore choose partners today who can not only deliver in the short term, but can provide regulatory support throughout the entire life cycle of a product. Inspital has embraced this change as an opportunity to further refine internal validation processes and elevate the reliability of every single component to a new level.
What is MDR? The New Gold Standard for Medical Devices
The Medical Device Regulation (MDR) has replaced the old Medical Device Directive (MDD) and imposes significantly stricter requirements on the approval and monitoring of medical devices in the EU. The main objective is to increase patient safety through complete traceability and stricter clinical evaluations. For products such as Inspital's operating tables or surgical lights, this means that every detail—from material properties to software control—is subject to the strictest testing cycles. A central aspect of the MDR is classification. While many hospital furniture items fall under Class I, more complex systems often require the involvement of "Notified Bodies." Inspital GmbH has already aligned its quality management system early on with the requirements of ISO 13485 and MDR (EU) 2017/745.
This guarantees our customers that they are investing in future-proof technology that will remain fully marketable beyond 2026. Beyond pure certification, this means increased legal certainty for the user in the event of liability issues. The stricter monitoring also ensures that inferior products are driven from the market, sustainably securing quality in European operating rooms. Inspital accompanies this process through transparent communication and ensures that all technical files are available for audits at all times.
EUDAMED and UDI: Maximum Transparency in the OR
One term most frequently searched in the context of the MDR is the EUDAMED database. From May 2026, the use of this European database for medical devices will be mandatory and fully operational. Related to this is the Unique Device Identification (UDI)—a barcode system that makes every medical device uniquely identifiable worldwide. Every component of an Inspital ceiling supply unit or surgical suction device receives this identification.
This offers hospitals enormous advantages in the areas of asset management and maintenance. Through UDI compliance, clinics can digitally track the life cycle of their Inspital products. In the event of a regulatory inquiry or necessary maintenance, all technical data are immediately retrievable. Inspital ensures that all documentation and labeling comply with the latest requirements to keep the administrative burden for hospital purchasers and medical technicians as low as possible. This digital traceability also drastically minimizes the risk of confusion with spare parts or accessories. Hospitals benefit from accelerated inventory management and a complete history of every device. For medical technology departments, this means significant relief in daily workflow and error-free documentation for regulatory authorities.
MDR: Clinical Evaluation and Post-Market Surveillance
Previously, an equivalence comparison with existing products was often sufficient to obtain approval. Under the MDR, a well-founded clinical evaluation is essential. Inspital continuously invests in the validation of its technologies. Whether it concerns the ergonomics of our motorized ceiling pendants or the light output of our LED surgical lights: we provide proof that our products deliver in clinical practice what they promise. Another cornerstone is Post-Market Surveillance (PMS). As a manufacturer, we are obligated to monitor our products throughout their entire life cycle in the market. Feedback from our users flows directly into product development.
When you choose stainless steel furniture or operating room equipment from Inspital, you participate in a continuous improvement process that extends far beyond the purchase. We see ourselves not only as a supplier, but as a partner for long-term safety. This systematic monitoring process enables us to identify potential risks before they become relevant in clinical practice. Our customers thus receive not just a static product, but a solution based on real application data and continuous scientific review. This significantly strengthens the trust of surgeons and nursing staff in the equipment they use daily.
Operating Tables and Surgical Lights: Safety Through Certified Quality
The heart of every operating room is the operating table and the surgical lighting. Under the MDR, these products are no longer viewed merely as mechanical aids. Particularly with motorized systems, electrical safety and electromagnetic compatibility (EMC) play a crucial role. Inspital's operating table systems are designed to offer maximum flexibility for various surgical disciplines while meeting all regulatory safety factors. Our surgical lights use state-of-the-art LED technology that not only provides excellent color rendering, but also meets strict requirements for heat generation and fail-safety.
Every device undergoes extensive testing procedures at Inspital in Ratingen before being deployed. By complying with MDR requirements, we ensure that medical personnel can work under optimal conditions without having to worry about technical conformity. The precise adjustability of our tables and the shadow-free illumination of our lamps are directly linked to the usability requirements of the MDR. We document in detail how our ergonomic designs reduce the burden on the team and thus lower the error rate. Every investment in Inspital technology is therefore also an investment in the ergonomic health of your staff and the safety of your patients.
Ceiling Supply Units: Modularity Meets Compliance
Planning an OR requires the highest precision. Ceiling supply units (CSU) from Inspital are complex systems that bundle gas connections, power supply, and data lines. Under the MDR, particularly strict rules apply to these combined systems regarding installation and interfaces. Our modular CSU solutions, such as the FX series, are designed to optimize workflows while maximizing hygiene requirements. Because our units do not occupy floor space, cleaning is facilitated—an essential factor for infection prevention, which is also indirectly addressed in the MDR through requirements for usability. Inspital offers turnkey solutions here that overcome all regulatory hurdles from planning to installation. Our experts support you in integrating the supply units perfectly into the existing infrastructure, always taking into account the latest standards. Particularly important here is the consideration of electromagnetic fields within the compact supply heads to exclude interference with sensitive monitoring equipment. Our engineers ensure that every individual configuration complies with strict EMC directives. Through this meticulous pre-planning, we avoid costly modifications during commissioning and guarantee a smooth acceptance process by technical inspection authorities.
Stainless Steel Furniture and Suction Devices: Hygiene as a Regulatory Requirement
In medical technology, the importance of seemingly "simple" products such as stainless steel hospital furniture is often underestimated. But the MDR also applies here: materials must be biocompatible and resistant to aggressive disinfectants. Inspital uses high-quality stainless steel specifically developed for the requirements of sterile environments. Our instrument tables, sinks, and cabinets are designed so that there are no "dead corners" where germs could settle.
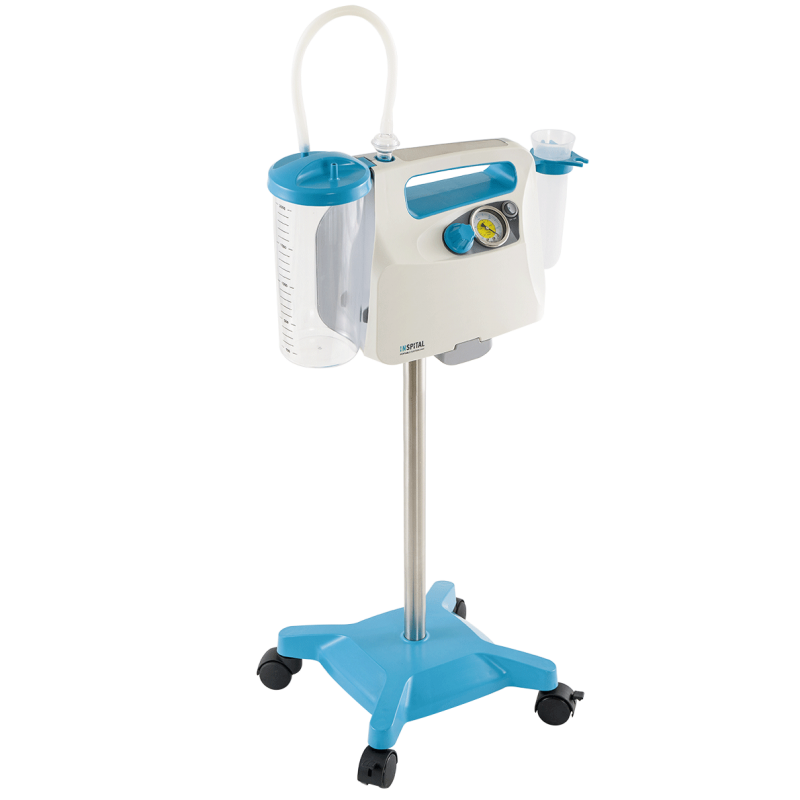
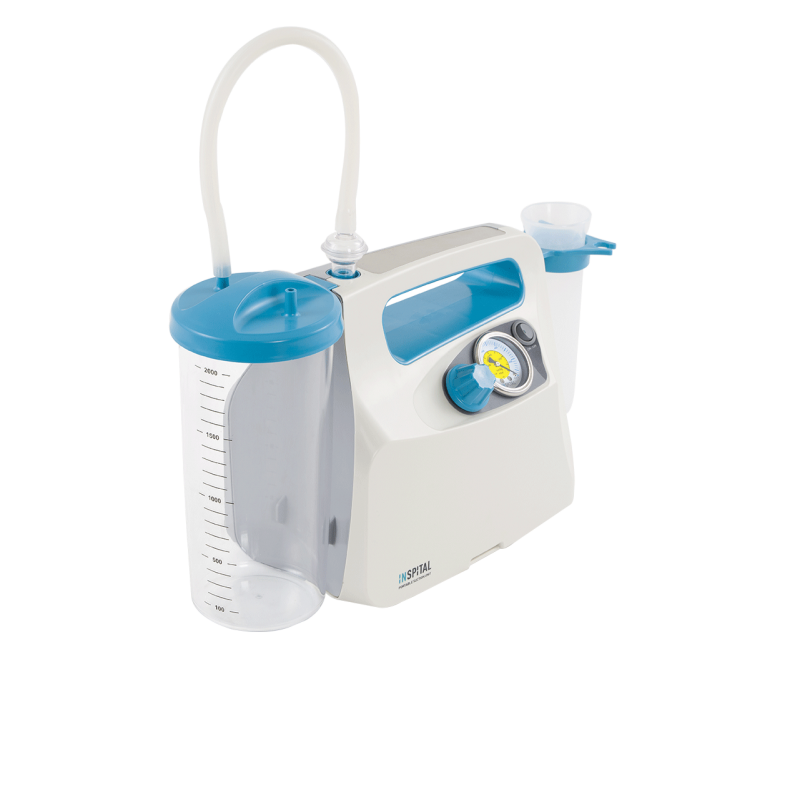
In addition, our portfolio includes surgical suction devices that are critical to patient safety during procedures. These devices must deliver precise performance data and be easy to maintain. In accordance with MDR requirements, we provide detailed technical documentation for all these products. This facilitates the creation of hospitals' own operator documentation and ensures legal certainty during audits by health authorities. Our suction devices are also characterized by intuitive operation, which minimizes the risk of user errors in stressful situations—a core requirement of the MDR for usability. Even with stainless steel furniture, we pay attention to rounded edges and seamless welding to minimize the risk of injury to personnel and accelerate sterilization processes. Thus, a piece of furniture becomes an active contribution to infection control.
Conclusion: Why Inspital Is Your Partner for the MDR Era
The transition to the MDR is a challenge for many hospitals, particularly regarding the selection of the right suppliers. Inspital GmbH has proactively shaped the transformation process. We offer not only products that are "MDR-ready," but comprehensive expertise for the operating room. From the first sketch to annual maintenance, we stand for German engineering and European safety standards. When you invest in medical technology, you invest in the future of your hospital. With Inspital, you choose a partner who understands the complexity of regulation and translates it into simple, efficient, and safe solutions. Trust in certified quality—for your patients, your staff, and your success. Our consultants in Ratingen are ready to develop customized concepts with you that not only shine today, but also withstand the requirements of tomorrow. We actively support you in evaluating your existing equipment and strategically replacing it with MDR-compliant new investments. Together, we create an operating room that is technologically leading and regulatorily unassailable. Contact us directly for a no-obligation consultation on your next project.
Operating Table OT80.20 NOVUS
Prices visible to registered customers onlySurgical Suction Machine SU60.05
Prices visible to registered customers onlySurgical Suction Unit SU60.10
Prices visible to registered customers onlyPortable Suction Unit SU60.06T
Prices visible to registered customers onlyCurrent News
- Current News, Hospital Management, Suction Units
- Current News, Hospital Management, Suction Units
- Current News, Hospital Management, Operating Table
News Press
Here you will find current news about trade fairs, congresses, PR and other relevant topics.